Instruction Manual
36 Pages
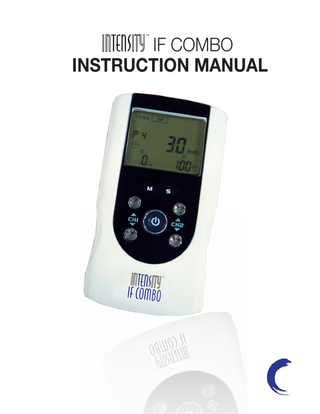
Preview
Page 1
TM
This manual is valid for the InTENSity Combo TENS and IF Stimulator
IF
This user manual is published by Current Solutions™, LLC Current Solutions™, LLC does not guarantee its contents and reserves the right to improve and amend it at any time without prior notice. Amendments may however be published in new editions of this manual. All Rights Reserved. Rev. V1.1 © 2010
: United States Federal Law restricts this device to sale by or on the order of a physician or licensed practitioner
Conformity to safety standards Current Solutions™, LLC declares that the device complies with following normative document: IEC60601-1, IEC60601-1-2, IEC60601-2-10, IEC60601-1-4, ISO10993-5, ISO10993-10, ISO10993-1
Table of Contents 4 1.SAFETY INFORMATIO 1.1 General description 1.2 Medical background 1.3 Indication for use 1.4 Contraindications 1.5 Warnings, Cautions, Adverse Reactions 2.PRESENTATION……… 2.1 Front and Rear panel 2.2 LCD display 3.SPECIFICATION 3.1 Accessories 3.2 Technical information 3.3 The waveforms of the stimulation programs 4.INSTRUCTION FOR U 4.1 Battery 4.2 Connect electrodes to lead wires 4.3 Connect lead wires to device 4.4 Electrodes 4.5 Turn ON 4.6 Select the Therapeutic Mode 4.7 Steps to set a new program 4.8 Adjust Channel Intensity 4.9 Safety Lock Feature 4.10 Stop the treatment 4.11 Turn OFF 4.12 Low battery indicator 5.PROGRAM……………………………………………………….……23 6. …………………………………….……...24 6.1 Tips for skin care 6.2 Cleaning the device 6.3 Electrodes 6.4 Cleaning the Electrode cords 6.5 Maintenance 7.TROUBLESHOOTIN 8.STORAGE………………………… 9. 10.ELECTROMAGNETIC COMPATIB 11.GLOSSARY OF SYMBLOS
1. Safety information 1.1 General InTENSity TM IF Combo stimulator is a portable electrotherapy device featuring two therapeutic modes: Transcutaneous Electrical Nerve Stimulation (TENS) and Interferential (IF), which are used for pain relief. The stimulator sends gentle electrical current to underlying nerves and muscle group via electrodes applied on the skin. The parameters of device are controlled by the buttons on the front panel. The intensity level is adjustable according to the needs of patients. 1.2 Medical background EXPLANATION OF PAIN Pain is a warning system and the body’s method of telling us that something is wrong. Pain is important; without it abnormal conditions may go undetected, causing damage or injury to vital parts of our bodies. Even though pain is a necessary warning signal of trauma or malfunction in the body, nature may have gone too far in its design. Aside from its value in diagnosis, long-lasting persistent pain serves no useful purpose. Pain does not begin until coded message travels to the brain where it is decoded, analyzed, and then reacted to. The pain message travels from the injured area along the small nerves leading to the spinal cord. Here the message is switched to different nerves that travel up the spinal cord to the brain. The pain message is then interpreted, referred back and the pain is felt. EXPLANATION OF TENS Transcutaneous Electrical Nerve Stimulation (TENS) is a noninvasive, drug free method of controlling pain. TENS uses tiny electrical impulses sent through the skin to nerves to modify your pain perception. TENS does not cure any physiological problem; it only helps control the pain. TENS does not work for everyone; however, in most patients it is effective in reducing or eliminating the pain, allowing for a return to normal activity. HOW TENS WORKS There is nothing “magic” about Transcutaneous Electrical Nerve Stimulation (TENS). TENS is intended to be used to relieve pain. The TENS unit sends comfortable impulses through the skin that stimulate the nerve (or nerves) in the treatment area. In many cases, 4
this stimulation will greatly reduce or eliminate the pain sensation the patient feels. Pain relief varies by individual patient, mode selected for therapy, and the type of pain. In many patients, the reduction or elimination of pain lasts longer than the actual period of stimulation (sometimes as much as three to four times longer). In others, pain is only modified while stimulation actually occurs. You may discuss this with your physician or therapist. EXPLANATION OF IF Interferential Stimulation (IF) is an anti-inflammatory based treatment modality. Interferential stimulation is characterized by two alternating-current sine waves or square waves of differing frequencies that “work” together to produce an interferential current that is also known as a beat pulse or alternating modulation frequency. One of the two currents is usually held at 4,000 Hz, and the other can be held constant or varied over a range of 4,001 to 4,100 Hz. Because of the frequency, the interferential wave meets low impedance when crossing the skin to enter deep into soft tissues. The interferential currents reportedly can stimulate sensory, motor, and pain fibers. These large impulse fibers interfere with the transmission of pain messages at the spinal cord level. This deep tissue penetration stimulates parasympathetic nerve fibers for increased blood flow and edema reduction. It utilizes the low electriccurrent to stimulate muscle nerves to achieve the symptomatic relief of chronic intractable pain, post-traumatic pain, and post-surgical pain. 1.3 Indication for use InTENSity IF Combo Stimulator may be used for the Symptomatic relief of chronic intractable pain, acute post traumatic pain or acute post surgical pain. TM
IMPORTANT SAFETY INFORMATION! Read instruction manual before operation. Be sure to comply with all “Contraindications”, Warnings”, “Cautions” and “Adverse reactions” in the manual. Failure to follow instructions can cause harm to user or device. 1.4 Contraindications 1 This device should not be used for symptomatic local pain relief unless etiology is established or unless a pain syndrome has been diagnosed. 5
This device should not be used on patients with epilepsy. This device should not be used on patients with serious arterial circulatory problems in the lower limbs This device should not be used on patients with abdominal or inguinal hernia
1.5 Warnings, Cautions and Adverse Reactions WARNINGS: of a licensed physician. 2) The long-term effects of chronic electrical stimulation are unknown. Electrical stimulation devices do not have any curative value. 3) TENS is a symptomatic treatment and, as such, suppresses the sensation of pain, which would otherwise serve as a protective mechanism. 4) Safety has not been established for the use of therapeutic electrical stimulation during pregnancy. Do not use during pregnancy unless directed by your physician. 5) Electrical stimulation is not effective for pain of central origin. 6) Electronic monitoring equipment (such as ECG monitors and ECG alarms) may not operate properly when electrical stimulation is in use. 7) Stimulation should not be applied over the carotid sinus nerves, particularly in patients with a known sensitivity to the carotid sinus reflex. 6
CAUTIONS: 1) Federal law (USA) restricts this device to sale by or on the order of a physician. 2) For single patient use only. 3) Keep yourself informed of the contraindications.
17) The electrodes are only to be placed on healthy skin. Avoid skin irritation by ensuring that good contact is achieved between electrodes and skin. 18) If the stimulation levels are uncomfortable or become uncomfortable, reduce the stimulation Intensity to a comfortable level and contact your physician if problems persist. 19) This device should not be used while driving, operating machinery, close to water, or during any activity in which involuntary muscle contractions may put the user at undue risk of injury. 20) Never use the device in rooms where aerosols (sprays) are used or pure oxygen is being administered. 21) Do not use it near any highly flammable substances, gases or explosives. 22) Do not use this device at the same time as other equipment which sends electrical pulses to your body. 23) Do not confuse the electrode cables and contacts with your headphones or other devices, and do not connect the electrodes to other devices. 24) Do not use sharp objects such as pencil point or ballpoint pen to operate the buttons on the control panel. 25) Inspect Applicator cables and associated connectors before each use. 26) Turn the device off before applying or removing electrodes. 27) Electrical stimulators should be used only with the leads and electrodes recommended for use by the manufacturer 28) This device has no AP/APG protection. Do not use it in the presence of explosive atmosphere and flammable mixture. Adverse Reactions: 1) Skin irritation from the electrode gel and electrode burns are potential adverse reactions. If skin irritation occurs, discontinue use and consult your physician. Note: Always use electrodes that are legally marketed and sold in the United States under 510K guidelines. 2) If the stimulation levels are uncomfortable, reduce the stimulation Intensity to a comfortable level and contact your physician if problems persist.
9
2. Presentation 2.1 Front and Rear Panel
1) Output socket: electric signal output after connection of the cable with adhesive electrodes channel 1. 2) Output socket: electric signal output after connection of the cable with adhesive electrodes channel 2. 3) Therapeutic mode selection (M). Stop the treatment. Exit setting mode to the user interface. 4) Increasing the output intensity of channel 1 [▲]. To set the application program and the parameter of the waveform in the setting state. 5) Decreasing the output intensity of channel 1 [▼]. To set the application program and the parameter of the waveform in the setting state. To unlock the current treatment program. 6) LCD display: Shows the operating state of the device. 7) Parameter Selection (S): press the button to enter setting state; you can select the difference parameters in conjunction with [▲] and [▼].
▲]. To set the ] button and hold for ▼]. To set the
2.2 LCD display
3. Specification 3.1 Accessories No 1 2 3 4 5 6 7
DESCRIPTION Electrical stimulator device Electrodes Leads 1.5” x 1.5” Adhesive Electrodes 9V Alkaline Battery, type 6LR61 Instruction Manual Carrying case AC Adaptor (optional)
Q’TY 1 piece 2 pieces 4 pieces 1 piece 1 piece 1 piece 1 piece
3.2 Technical information Channel Power supply Operating conditions Storage conditions Dimensions Weight Tolerance Timer
Electrode Detection Function
Dual, isolated between channels 9.0 V Alkaline DC -1 *6LR61 battery Adapter output:9.0Vdc 800mA 5°C to 40°C (41℉ to 104℉)with a relative humidity of 30%-75%,atmospheric pressure from 700 to 1060 Hpa -10°C to 50°C (14℉ to 122℉)with a relative humidity of 10%-90%,atmospheric pressure from 700 to 1060 Hpa 4.5×2.55×0.9 inches(L*W*H) 0.28 lbs(With battery) There may be a ±5% tolerance of all setting and ±10% tolerance of output of intensity. Adjustable, from 1 to 60 minutes or continuous, Adjustable in 1minutes each step. Treatment time countdown automatically. The amplitude level will be reset to 0mA when the amplitude level is 12mA or greater and an open circuit at either channel is detected.
12
Technical specifications for Transcutaneous Electrical Nerve Stimulator (TENS) mode Waveform
Mono-phase square pulse wave Adjustable, 0~105mA peak at 1000 Pulse amplitude ohm Load each channel, 1mA/Step. Adjustable, from 50 to 300us Pulse Width microseconds, 10μS/step Adjustable, from 1 to 150 Hz, 1 Pulse Rate Hz/step Burst rate: Adjustable, 0.5 ~ 5Hz; 0.1Hz/step Burst (P1) Pulse width adjustable, 50~300μS Frequency fixed = 100 Hz The pulse rate and pulse width are adjustable. It generates continuous Normal (P2) stimulation based on the setting value. The pulse width is automatically varied in a cycle time. The pulse width is decreased from its original setting to 60% in setting cycle time, Pulse Width Modulation and then increased from 60% to its (P3) original setting in nest setting cycle time. In this program, pulse rate (1 to 150Hz), pulse width (50 to 300us) and cycle time (5 to 30 sec) are fully adjustable. The pulse rate is automatically varied in a cycle time. The pulse rate is decreased from its original setting to 60% in setting cycle time, and then increased from 60% to its Pulse Rate Modulation (P4) original setting in nest setting cycle time. In this program, pulse rate (1 to 150Hz), pulse width (50 to 300us) and cycle time (5 to 30 sec) are fully adjustable.
13
Technical specifications for Interferential (IF) mode Waveform Pulse amplitude Pulse Rate
Phase Width
P1
P2
P3
Bi-phase square pulse Adjustable, 0~70mA peak to peak at 1000 ohm Load each channel, 1mA/Step. Channel 1 – Fundamental frequency: 4000 Hz fixed Channel 2 – Selectable frequency: 4001 to 4150 Hz Interference frequency: 1 to 150 Hz. 125μs The pulse rate of the CH1 is fixed in 4000Hz; CH2 pulse rate is increased from 4001Hz to 4010Hz in a cycle time, and then decreased from 4010Hz to 4001Hz in nest setting cycle time. In this program, CH2 interference frequency is varied from 1Hz to 10Hz, cycle time (5 to 30 sec) is fully adjustable. CH 2 pulse rate=4000Hz+ Interference frequency The pulse rate of the CH1 is fixed in 4000Hz; CH2 pulse rate is increased from 4001Hz to 4150Hz in a cycle time, and then decreased from 4150Hz to 4001Hz in nest setting cycle time. In this program, CH2 interference frequency is varied from 1Hz to 150Hz, cycle time (5 to 30 sec) is fully adjustable. CH 2 pulse rate=4000Hz+ Interference frequency The pulse rate of the CH1 is fixed in 4000Hz; CH2 pulse rate is increased from 4080Hz to 4150Hz in a cycle time, and then decreased from 4150Hz to 4080Hz in nest setting cycle time. In this program, CH2 interference frequency is varied from 80Hz to 150Hz, cycle time (5 to 30 sec) is fully adjustable. CH 2 pulse rate=4000Hz+ Interference frequency 14
P4
The pulse rate of the CH1 is fixed in 4000Hz; CH2 pulse rate is automatically varied in a cycle time. Interference frequency is increased from its original setting to 60% in setting cycle time, and then decreased from 60% to its original setting in nest setting cycle time. In this program, CH2 interference frequency (2 to 150Hz) and cycle time (5 to 30 sec) are fully adjustable. CH 2 pulse rate=4000Hz+ Interference frequency
3.3 The waveforms of the stimulation programs Burst
Normal
Pulse Width Modulation
15
Pulse Rate Modulation
Interferential
4. Instruction for use 4 .1 Battery 4.1.1 Check/Replace the battery Over time, in order to ensure the functional safety of device, changing the battery is necessary. 1) Slide the battery compartment cover and open. 2) Insert the 9V battery into the battery compartment. 3) Make sure you are installing the battery properly. Be sure to match the positive and negative ends of the battery to the marking in the battery compartment of the device. 4) Press and pull down following the direction of the arrow indicated on the photo. 5) Replace the battery compartment cover and press to close 6) If replace the battery, you should slide the battery compartment cover and open. Pull up the battery following the direction of the arrow indicated on the photo. And insert the 9V battery according to the above steps 2-5. 16
4.1.2 Disposal of battery Spent batteries do not belong in the household waste. Dispose of the battery according to the current federal, state and local regulations. Caution: 1) Battery may be fatal if swallowed. Therefore, keep the battery and the product out of the range of children, if a battery was swallowed, consult a physician immediately. 2) If a battery has leaked, avoid contact with skin, eyes and mucus membranes, Rinse the affected spots with lots of clear water immediately and contact a physician right away. 3) Battery may not be charged, dismantled, thrown into fire or short-circuited. 4) Protect battery from excess heat; Take the battery out of the product if they are spent or in case you no longer use the article. This prevents damage caused by leaking battery. 5) Always replace the same type battery.
4.2 Connect electrodes to lead wires Insert the lead wire connector into electrodes connector (standard 0.08 inch female connection). Make sure no bare metal of the pins is exposed. Caution: Always use the electrodes with the requirements of the IEC/EN60601-1 ,ISO10993-1/-5/-10 and IEC/ EN60601-1-2, such as with CE mark, or which are legally marketed in the US under 510(K) procedure.
4.3 Connect lead wires to device 1) Before proceeding to this step, be sure the device is completely turns OFF.
17
2) The wires provided with the system insert into the jack sockets located on top of the device. 3) Holding the insulated portion of the connector, push the plug end of the wire into one of the jacks (see drawing); one or two sets of wires may be used. 4) This device has two output receptacles controlled by Channel 1 and Channel 2 at the top of the unit. You may choose to use one channel with one pair of lead wires or both channels with two pairs of lead wires. Using both channels gives the user the advantage of stimulating two different areas at the same time. Caution: Do not insert the plug of the patient lead wire into any AC power supply socket.
4.4 Electrode 4.4.1 Electrode options The electrodes are disposable and should be routinely replaced when they start to lose their adhesive nature. If you are unsure of your electrode adhesive properties, order replacement electrodes. Replacement electrodes should be re-ordered through or on the advice of your physician to ensure proper quality. Follow application procedures outlined in electrode packing, to maintain optimal stimulation and to prevent skin irritation. 4.4.2 Place electrodes on skin Apply electrodes to the exact site indicated by your physician or therapist, before applying electrodes, be sure the skin surface over which electrodes are placed is thoroughly cleaned and dried. Make sure the electrodes are placed firmly to the skin and make good contact between the skin and the electrodes. Place the electrodes over the skin; attach them properly, firmly, and evenly. 18
Caution: 1) Before applying the self-adhesive electrodes, it is recommended to wash and degrease the skin, and then dry it. 2) Do not turns on the device when the self-adhesive electrodes are not positioned on the body. 3) Never remove the self-adhesive electrodes from the skin while the device is still turns on. 4) It is recommended that, at minimum 1.5" x 1.5" self-adhering based, square electrodes are used at the treatment area 4.4.3 Electrode placement The placement of electrodes can be one of the most important parameters in achieving success with therapy. Of utmost importance is the willingness of the physician to try the various styles of electrode placement to find which method best fits the needs of the individual patient. Every patient responds to electrical stimulation differently and their needs may vary from the conventional settings suggested here. If the initial results are not positive, speak to your physician about alternative stimulation settings and/or electrode placements. Once an acceptable placement has been achieved, mark down the electrodes sites and the settings, so the patient can easily continue treatment. 4.5 Turn on Before using the device for the first time, you are strongly advised to take careful note of the contraindications and safety measures detailed at the beginning of this manual (Safety information), as this powerful equipment is neither a toy nor a gadget ! In order to turn on the devi The operation page appears on the screen. 4.6 Select the Therapeutic Mode There are two therapeutic modes available –TENS and IF. The therapeutic mode can be selected by pressing the [M] control.
Caution: Consult your physician for your suitable therapeutic mode 4.7 Steps to Set a New Program 4.7.1 TENS Setting Press the [S]button cycle to enter the setting state. The settings can be adjusted according to the following steps: 1) Set the Therapeutic Program There are 4 programs in TENS therapeutic mode available –Burst (P1), Normal (P2), Pulse Width Modulation (P3), and Pulse Rate Modulation (P4). The therapeutic program can be selected by pressing the [▲] and [▼] button. 2) Set Cycle Time (Optional) Cycle time is adjustable form 5 to 30 seconds. Only modulation has this parameter setting. Press [S] button cycle to enter this menu, and then press the [▲] and [▼] button to adjusting the setting. 3) Set Timer Press [S] button cycle to enter this setting. The treatment time is adjustable from 1 to 60 minut control to adjust setting. You can set the timer to “Continuous” mode by pressing the [▲] control when it shows 60 minutes. Its output will be shut off when time is up. 4) Set Pulse Width Pulse Width is adjustable from 50 us to 300 us. Press [S] button to enter this menu, then press [▲] or [▼] button to adjust the setting. 5) Set Pulse Rate Pulse rate is adjustable from 1 Hz to 150 Hz (0, 5 Hz to 5 Hz for Burst). Press [S] button cycle to enter this menu, and then press [▲] or [▼] button to adjust the setting.
4.7.2 IF Setting Press the [S] button to enter the setting state. The settings can be adjusted according to the following steps: 1) Set the Therapeutic Program There are 4 programs in IF therapeutic mode available. The therapeutic program can be selected by pressing the [▲] and [▼] button. The mode you selected will show up on the top of liquid crystal display.
2) Set Timer Press [S] button cycle to enter this setting. The treatment time is adjustable from 1 to 60 minutes or Continuous. Press [▲] or [▼] control to adjust setting. You can set the timer to “Continuous” mode by pressing the [▲] button when it shows 60 minutes. Its output will be shut off when time is up. 3) Set Interference frequency (optional) Channel 1 has 4000 Hz fixed Fundamental frequency. Channel has selectable frequency from 4001 to 4150 Hz; Interference frequency is adjustable form 1 Hz to 150 Hz. Only “P4” has this parameter setting. Press “S” button cycle to enter this menu, and then press the [▲] and [▼] button to adjusting the setting. 4) Set Cycle Timer Cycle time is adjustable form 5 to 30 seconds. Press [S] button cycle to enter this menu, and then press the [▲] and [▼] button to adjusting the setting.
4.8 Adjust Channel Intensity Press the intensity control button ([▲] and [▼]) to control the intensity output. Slowly press the intensity button control until you reach the setting recommended by your physician or therapist. Repeat for the other channel, if both channels are to be used. Caution: 1) If the stimulation levels are uncomfortable or become uncomfortable, reduce the stimulation intensity to a comfortable level and contact your medical practitioner if problems persist. 2) If the electrodes no placed firmly on skin or the device has not connected on the electrodes, the stimulator’s output intensity surpasses 12mA, the intensity will enulls automatically reset to 0mA. 4.9. Safety Lock Feature The Safety Lock Feature automatically activates after there is no operation in the panel for 30 seconds by locking out the ability to press the buttons. This is a safety feature to prevent accidental changes to your settings and to prevent accidental increases to the intensity levels. You can press either one of the [▼] buttons to unlock the device. 4.10. Stop the treatment When you have activated the treatment timer, you can press the [M] button or the [▼] button to control stop the treatment. Caution: Default state; if the button is locked, you can press only one of the [▼] buttons to unlock, and then press the [M]button or the [▼] button to control stop the treatment.
22