Technical Manual
245 Pages
Preview
Page 1
Rendered PDF File Page 1 of 245
DOC0951990, Rev:3
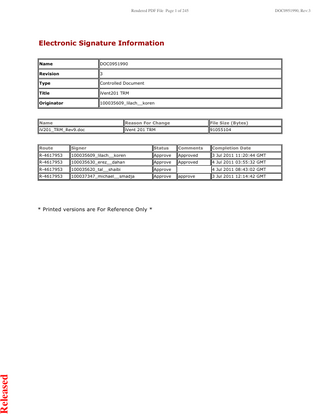
Electronic Signature Information Name
DOC0951990
Revision
3
Type
Controlled Document
Title
iVent201 TRM
Originator
100035609_lilach__koren
Name
Reason For Change
File Size (Bytes)
iV201_TRM_Rev9.doc
iVent 201 TRM
91055104
Route
Signer
Status
Comments
Completion Date
R-4617953
100035609_lilach__koren
Approve
Approved
3 Jul 2011 11:20:44 GMT
R-4617953
100035630_erez__dahan
Approve
Approved
4 Jul 2011 03:55:32 GMT
R-4617953
100035620_tal__shaibi
Approve
R-4617953
100037347_michael__smadja
Approve
Released
* Printed versions are For Reference Only *
4 Jul 2011 08:43:02 GMT approve
3 Jul 2011 12:14:42 GMT
Rendered PDF File Page 2 of 245
TM
i Vent
201
Technical Reference Manual
iVentTM201 Technical Manual VersMed Part Number SM-01-04 GE Part Number M1162065 Rev: 6 Revised April 2011 / 19.18.02 Copyright © 2011 by GE Medical Systems Israel Ltd – VersaMed Business
Released
0473
DOC0951990, Rev:3
Rendered PDF File Page 3 of 245
Owner’s Record
The model number and serial number of your iVentTM201 are on the rear panel of your ventilator. Record the serial number in the space provided below to have this information should you need to call for service or support. Model Number:
Serial Number:
iVentTM201 is a registered trademark of GE Medical Systems Israel Ltd – VersaMed Business Other brand names or product names used in this manual are trademarks or registered trademarks of their respective holders.
Obelis S.A. Av. de Tervuren 34, bte 44 B-1040 Brussels Belgium
Released
Tel: +32-2-732-59.54 Fax: +32-2-732-60.03
DOC0951990, Rev:3
Released
Rendered PDF File Page 4 of 245
DOC0951990, Rev:3
Rendered PDF File Page 5 of 245
Important The information contained in this Technical Reference manual pertains only to those models of products which are marketed by VersaMed as of the effective date of this manual or the latest revision thereof. This Technical Reference manual was prepared for exclusive use by VersaMed service personnel in light of their training and experience as well as the availability to them of parts, proper tools and test equipment. Consequently, VersaMed provides this Technical Reference manual to its customers purely as a business convenience and for the customer's general information only without warranty of the results with respect to any application of such information. Furthermore, because of the wide variety of circumstances under which maintenance and repair activities may be performed and the unique nature of each individual's own experience, capacity, and qualifications, the fact that customer has received such information from VersaMed does not imply in any way that VersaMed deems said individual to be qualified to perform any such maintenance or repair service. Moreover, it should not be assumed that every acceptable test and safety procedure or method, precaution, tool, equipment or device is referred to within, or that abnormal or unusual circumstances, may not warrant or suggest different or additional procedures or requirements. This manual is subject to periodic review, update and revision. Customers are cautioned to obtain and consult the latest revision before undertaking any service of the equipment. Comments and suggestions on this manual are invited from our customers. Send your comments and suggestions to the Manager of Technical Communications, Datex-Ohmeda, Ohmeda Drive, PO Box 7550, Madison, Wisconsin 53707.
CAUTION Servicing of this product in accordance with this Technical Reference manual should never be undertaken in the absence of proper tools, test equipment and the most recent revision to this service manual which is clearly and thoroughly understood.
Technical Competence The procedures described in this Technical Reference manual should be performed by trained and authorized personnel only. Maintenance should only be undertaken by competent individuals who have a general knowledge of and experience with devices of this nature. No repairs should ever be undertaken or attempted by anyone not having such qualifications. VersaMed strongly recommends using only genuine replacement parts, manufactured or sold by VersaMed for all repair parts replacements.
Released
Read completely through each step in every procedure before starting the procedure; any exceptions may result in a failure to properly and safely complete the attempted procedure.
DOC0951990, Rev:3
Released
Rendered PDF File Page 6 of 245
DOC0951990, Rev:3
Released
Rendered PDF File Page 7 of 245
DOC0951990, Rev:3
Released
Rendered PDF File Page 8 of 245
DOC0951990, Rev:3
Rendered PDF File Page 9 of 245
Table of Contents Section 1: Introduction ...1 1.1 Intended Use ... 1 1.2 General Description... 2 1.2.1 Looking At The iVentTM201 ... 3 1.2.2 Product Versions ... 5 1.2.3 Safety Information... 6 1.2.4 Safety Regulations ... 7 1.2.5 Handling PC Boards ... 8 1.3 Labels and Symbols ... 9 1.3.1 Symbols ... 9 1.3.2 Labels ... 11
Section 2: System Specifications ...15 2.1 Ventilation Modes ... 15 2.2 Ventilation Performance and Controlled Parameters ... 15 2.3 Monitored Data ... 17 2.4 Monitoring and Displayed Parameters... 20 2.5 Adjustable Non-Displayed Parameters... 21 2.6 User Adjustable Alarms ... 22 2.7 Additional Alarms... 22 2.8 Size and Weight ... 23 2.9 Power Supply... 23 2.10 Oxygen Supply... 23 2.11 Environmental Specifications ... 24
Released
2.12 Standards and Safety Requirements ... 24
DOC0951990, Rev:3
Rendered PDF File Page 10 of 245
2.13 Indicators and Icons ...25
Section 3: Installation and Setup... 26 3.1 Introduction ...26 3.2 Unpacking the Ventilator ...26 3.3 Connection of Power Sources...26 3.3.1 External Power Sources ...26 3.3.2 Internal Power Pack...28 3.3.3 Electrical Safety Testing ...29 3.4 Connecting Alternate Power Sources ...29 3.4.1 High Pressure Oxygen...29 3.4.2 Low Pressure Oxygen ...31 3.5 Patient Circuit and Accessory Connection ...32 3.6 Patient Circuit...32 3.6.1 Disposable Patient Circuit...33 3.6.2 Reusable Patient Circuit...34 3.6.3 Dual Limb Patient Circuit ...34 3.6.4 Circuit Resistance ...36 3.7 Pre Use Testing...37 3.8 Filters...37 3.8.1 Air Inlet Filter ...37 3.9 Ventilator Controls ...40 3.9.1 Rotational Control Knob (Encoder) ...40 3.9.2 Keypad ...40 3.9.3 LED Indicators...41 3.10 Ventilator Operation ...41
Released
Section 4: Theory of Operation ... 42
DOC0951990, Rev:3
Rendered PDF File Page 11 of 245
4.1 Overview of System Operation ... 42 4.2 Pneumatic System ... 43 4.2.1 Inlet Filter and Manifold ... 48 4.2.2 Oxygen Blending System – PSV Units (iV15000>) ... 48 4.2.3 Oxygen Blending System - Stepper Units (<iV14999) ... 50 4.2.4 Turbine System... 52 4.2.5 Solenoid Valve System... 52 4.2.6 O2 Sensor ... 53 4.2.7 Patient Connector & Outlet Muffler... 54 4.2.8 Cooling Fan... 55 4.2.9 Patient Circuit... 55 4.3 Electronic System ... 58 4.3.1 Power Entry Module... 61 4.3.2 Power Supply PCB... 61 4.3.3 Integrated Power Pack... 61 4.3.4 External DC Supply... 62 4.3.5 Switching PCB... 62 4.3.6 CPU PCB ... 65 4.3.7 Main Board... 68 4.3.8 Zeroing/Purge Board ... 73 4.3.9 Hold/Nebulizer Controller PCB... 73 4.3.10 Mounting PCB... 73 4.3.11 Switching Board... 74 4.3.12 Power Supply (AC/DC Converter)... 77 4.4 LCD Display ... 77
Released
4.5 Interface Board... 78
DOC0951990, Rev:3
Rendered PDF File Page 12 of 245
Section 5: Maintenance Menu ... 79 5.1 Introduction ...79 5.2 Accessing the Maintenance Menu...79 5.3 Maintenance Menu ...80 5.4 Calibration Screen ...80 5.5 Ventilator Verification Test ...80 5.6 Configuration Screen...81 5.6.1 Software Version ...81 5.6.2 Default Start Screen ...81 5.6.3 Default Start Weight...82 5.6.4 Default FIO2 Setting ...82 5.6.5 Serial Number ...82 5.6.6 Card Number...83 5.6.7 Sensor Type...83 5.6.8 Package Type...83 5.7 Service Screen ...84 5.7.1 Communication Port ...84 5.7.2 Communication Rate ...84 5.7.3 Update Software Version...85 5.7.4 Update Package Key ...85 5.7.5 Reset Battery LMD...86 5.8 Technical Log Book ...87 5.9 Localization ...88 5.9.1 Language ...89 5.9.2 Date / Interfield Format...89
Released
5.10 Total Operation Hours...90
DOC0951990, Rev:3
Rendered PDF File Page 13 of 245
Section 6: Self-Tests ...91 6.1 Introduction... 91 6.2 Operator Verification Test... 91 6.2.1 O.V.T. Requirements... 91 6.2.2 Accessing O.V.T. ... 91 6.2.3 O.V.T. Procedure: ... 91 6.2.4 O.V.T. Technical Description ... 93 6.3 Ventilator Verification Test ... 95 6.3.1 V.V.T. Requirements ... 96 6.3.2 Accessing V.V.T... 96 6.4 V.V.T. Procedure:... 96 6.4.1 V.V.T. Technical Description... 102 6.5 Troubleshooting Self-Tests ... 106 6.5.1 OVT Tests ... 106 6.5.2 VVT Tests... 107
Section 7: Calibrations...112 7.1 Introduction... 112 7.2 Calibration Overview... 112 7.3 When to Run Calibration ... 113 7.4 Calibration Requirements... 113 7.5 Accessing Calibration... 114 7.6 Calibration Procedures ... 114 7.6.1 Zero Sensors Calibration ... 114 7.6.2 Pressure Sensor Calibration ... 115
Released
7.6.3 PEEP-RPM Calibration... 117
DOC0951990, Rev:3
Rendered PDF File Page 14 of 245
7.6.4 Flow Sensor Calibration ...119 7.6.5 Volume Calibration...120 7.6.6 O2 System Calibration ...122 7.6.7 Saving Calibration Data...125 7.7 Battery Gas Gauge Initialization Procedure...126 7.8 Troubleshooting Calibrations ...127 7.8.1 Calibration Tests...127 THIS PAGE INTENTIONALL LEFT BLANK ...129
Section 8: Functional Test Procedure... 130 8.1 Introduction ...130 8.2 Notes ...130 8.3 When to Run Tests ...130 8.4 Test Equipment & Materials...131 8.5 Cleaning and Inspection ...131 8.6 Performance Testing ...132 8.6.1 Electrical Safety Test...132 8.6.2 Oxygen Performance Tests ...134 8.6.3 Alarm Tests ...137
Section 9: Planned Maintenance... 143 9.1 Introduction ...143 9.2 Cleaning and Routine Planned Maintenance ...143 9.2.1 Cleaning the Ventilator...144 9.3 Planned Maintenance ...144 9.3.1 500 Hour PM Procedure ...146
Released
9.3.2 1500 Hour PM Procedure ...146
DOC0951990, Rev:3
Rendered PDF File Page 15 of 245
9.3.3 3000 hours PM Procedures... 147 9.3.4 Annual PM Procedure... 147 9.3.5 24 Months ... 148 9.3.6 15000 Hour PM Procedure ... 148
Section 10: Software Applications ...149 10.1 Introduction... 149 10.2 Hardware Requirements ... 149 10.3 Software Utilities Set Up ... 149 10.4 Software Utility Availability... 150 10.5 Technical Error / Event Log Download Utility ... 150 10.6 Ventilator Software Upgrade Procedure ... 153 10.6.1 Installation Requirements... 155 10.6.2 Parts List for System Software 19.18.02 upgrade kit ... 155 10.6.3 Software Update ... 155 10.6.4 Checkout Procedure ... 162
Section 11: Service & Repair Procedures...164 11.1 Introduction... 164 11.2 Repair Safety ... 164 11.3 Repair Guidelines ... 165 11.4 Cleaning... 166 11.5 Electrical Cables and Pneumatic Connections ... 166 11.6 Electro-Static Discharge (E.S.D) Control... 166 11.6.1 General Information ... 166 11.6.2 Procedures and Precautions ... 167
Released
11.7 Non-Conforming Parts... 167
DOC0951990, Rev:3
Rendered PDF File Page 16 of 245
11.8 Replacement Parts ...168 11.9 Post Repair ...168 11.10 Repair Documentation ...168 11.11 Patient System and Accessory Devices...169 11.12 Recommended Service Tooling and Materials ...169 11.13 Service and Repair Procedures ...169 11.13.1 Enclosure Disassembly...169 11.13.2 Electronic Module Removal and Installation...174 11.13.3 Pneumatic Unit Removal and Installation ...177 11.13.4 Power Pack Disassembly and Assembly ...181 11.13.5 O2 Sensor Removal and Installation ...182
Section 12: Troubleshooting ... 186 12.1 Troubleshooting Guide...186 12.2 Diagnostics and Repairs...189 12.2.1 Demand Valve ...189 12.2.2 Pressure Switch ...191 12.2.3 Root Cause - Pneumatic vs. Electronic ...192 12.3 Exhale VT Accuracy...195 12.3.1 Inhale VT Accuracy ...195 12.3.2 Exhale VT Accuracy Interferences ...196 12.3.3 Leakage ...196 12.3.4 Velocity ...196 12.3.5 MAQUET (Siemens 190) Test lung ...197 12.4 Miscellaneous Issues ...198 12.4.1 Black Screen ...198
Released
12.4.2 Fails Calibrate Flow Sensor ...198
DOC0951990, Rev:3
Rendered PDF File Page 17 of 245
12.4.3 Erratic O2 Control... 198 12.4.4 Erratic Exhale Tidal Volumes ... 199 12.4.5 Low Pressure during Calibration or VVT ... 199 12.5 Setting Up the Ventilator for Static Pressure... 199
Section 13: Spare Parts and Accessories...201 13.1 Introduction... 201 13.2 Hardware Versions ... 201 13.3 Parts Lists... 202 13.3.1 Ventilator Accessories ... 202 13.3.2 Sub-Assemblies... 204 13.3.3 Rear Panel Components ... 206 13.3.4 Integrated Battery Components ... 207
Released
Index...217
DOC0951990, Rev:3
Rendered PDF File Page 18 of 245
DOC0951990, Rev:3
Illustrations Figure 1-1 Front View of the iVentTM201... 3 Figure 1-2 Rear View of the iVentTM201 ... 4 Figure 3-1 Power connections to iVentTM201 ... 27 Figure 3-2 oxygen DISS inlet connector... 30 Figure 3-3 Low pressure oxygen connector ... 31 Figure 3-4 Patient Circuit Connection ... 33 Figure 3-5 Patient Circuit Connections (Breathing Circuit)... 33 Figure 3-6 Exhalation valve adapter installation ... 35 Figure 3-7 iVentTM201 Filters ... 39 Figure 3-8 Ventilator Controls... 40 Figure 4-1 Pneumatic Unit – PSV Based System... 44 Figure 4-2 Pneumatic Unit – Stepper Based System... 45 Figure 4-3 Pneumatic System – PSV Based Units ... 46 Figure 4-4 Pneumatic System – Stepper Based Units ... 47 Figure 4-5 Hold/Nebulizer Mounting ... 57 Figure 4-6 Electronic Module ... 59 Figure 4-7 Ventilator Overview... 60 Figure 4-8 Remote alarm connection ... 72 Figure 5-1 Main Menu and Confirmation Screens ... 79 Figure 5-2 Maintenance Menu Screen ... 80 Figure 5-3 Configuration Screen ... 81 Figure 5-4 Service Screen ... 84 Figure 5-5 Package Key Update Screen ... 86 Figure 5-6 Battery type selection... 87 Figure 5-7 Confirmation window ... 87 Figure 5-8 Technical Error Log Screen... 88
Released
Figure 5-9 Localization Screen... 89
Rendered PDF File Page 19 of 245
Figure 6-1 O.V.T. part one...92 Figure 6-2 O.V.T part two ...92 Figure 6-3 Initiating V.V.T. ...96 Figure 6-4 Pressure Tests ...97 Figure 6-5 Pressure Test Results...98 Figure 6-6 Flow Tests ...98 Figure 6-7 O2 Tests (21%)...99 Figure 6-8 O2 Tests (100%)...99 Figure 6-9 Disconnect AC Power ... 100 Figure 6-10 AC Reconnect / Watchdog Test... 101 Figure 6-11 VVT Test Results... 101 Figure 7-1 Entering the Calibration Menu... 114 Figure 7-2 Zero Sensors... 115 Figure 7-3 Calibrate Pressure Sensors... 116 Figure 7-4 Calibrate PEEP-RPM ... 118 Figure 7-5 Calibrate Flow Sensor ... 119 Figure 7-6 Calibrate Volume... 121 Figure 7-7 O2 system calibration screens (Stepper Units)... 122 Figure 7-8 O2 Calibration Cover ... 124 Figure 7-9 O2 System Calibration Screens (Proportional Valve Units)... 124 Figure 7-10 Save Calibration... 126 Figure 8-1 Electrical Safety Test Set Up ... 133 Figure 8-2 Oxygen performance test setup ... 134 Figure 8-3 Internal O2 Measurement... 135 Figure 8-4 100% O2 (Suction) Mode ... 136 Figure 8-5 High Pressure Alarm... 137 Figure 8-6 Apnea Alarm... 138 Figure 8-7 Tube Disconnect Alarm ... 139 Figure 8-8 Patient Disconnect Alarm ... 140 Figure 9-1 Cooling Vent (left) and Cooling Air Inlet Filter (right) ... 147
Released
Figure 10-1 Software utilities cabling setup ... 150
DOC0951990, Rev:3
Rendered PDF File Page 20 of 245
DOC0951990, Rev:3
Figure 10-2 iVDownload Application Window... 152 Figure 10-3 Event Log ... 152 Figure 10-4 Error Log ... 153 Figure 10-5 Software Installation screen (for SW version 19.17.01 or 19.17.02) ... 156 Figure 10-6 Software Installation Screen ... 157 Figure 10-7 Software installation application ... 158 Figure 10-8 Software installation application – establish connection ... 158 Figure 10-9 Software installation application – Connection established ... 159 Figure 10-10 iVent maintenance menu ... 159 Figure 10-11 iVent Maintenance warning message... 160 Figure 10-12 iVent Service Screen ... 160 Figure 10-13 iVent is ready for upgrading ... 160 Figure 10-14 Installation completed message ... 161 Figure 10-15 iVent configuration screen... 162 Figure 11-1 Enclosure (Rear View) #1 ... 171 Figure 11-2 Front Enclosure (Inside View) #1 ... 172 Figure 11-3 Rear Enclosure (Inside View) #1... 172 Figure 11-4 Enclosure (Rear View) #2 ... 175 Figure 11-5 Rear Enclosure (Inside View) #2... 176 Figure 11-6 Rear Enclosure (Inside View) #3... 178 Figure 11-7 O2 Mixer Cam to Mid-Point Position... 179 Figure 11-8 PU Ground Point Locations... 180 Figure 11-9 Power Pack (Rear View) ... 182 Figure 11-10 Power Pack (Removed)... 182 Figure 11-11 The O2 sensor location on the pneumatic unit ... 183 Figure 11-12 The Shield holder 3 legs... 184 Figure 12-1 O2 Inlet Pipe and Demand Valve ... 191 Figure 12-2 PU Main Connector (Front View) - Pressure Switch Pinout ... 192 Figure 12-3 Flow Graphs (Occluding the Positive Flow Sensor Port)... 195 Figure 12-4 Flow Graphs (High Velocity Exhale)... 197
Released
Figure 12-5 Test Lung 190 and iVentTM201 Waveforms... 198